| Clinical Study "Common" Name (formal title) | Number of enrollees | Funding source (pharma/NIH) | Year complete | Primary publication | Full study treatment protocol |
|---|---|---|---|---|---|
|
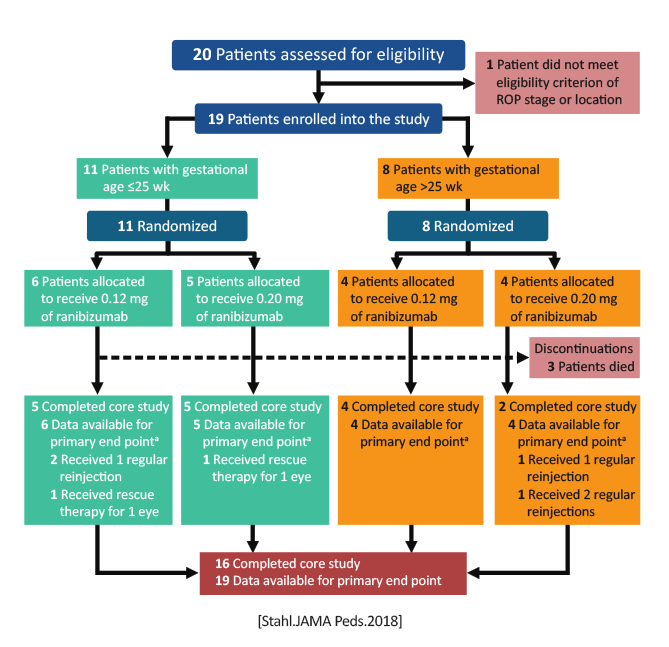
CARE-ROP [Multicenter Randomized Double Masked Parallel Design Exploratory Study to Assess Safety and Efficacy of Two Different Doses of Intravitreal Anti-VEGF Treatment With Ranibizumab (0.12 mg vs. 0.20 mg) in Infants With Retinopathy of Prematurity (ROP)] |
19 |
University Hospital Freiburg NCT02134457 and clinicaltrialsregister.eu Identifier: 2013-002539-13. |
Primary and study completion: January 2017 |
Stahl A, Krohne TU, Eter N, et al. Comparing alternative ranibizumab dosages for safety and efficacy in retinopathy of prematurity: A randomized clinical trial. JAMA Pediatr. 2018;172:278-286. doi.org/10.1001/jamapediatrics.2017.4838 |
 [Stahl.JAMA Peds.2018] [Stahl.JAMA Peds.2018]
|