| Clinical Study "Common" Name (formal title) | Study design | Treatment arms | Primary outcomes | Results | Author conclusions | Real-world impact |
|---|---|---|---|---|---|---|
|
BALATON [A Phase III, Multicenter, Randomized, Double-Masked, Active Comparator-Controlled Study to Evaluate the Efficacy and Safety of Faricimab in Patients With Macular Edema Secondary to Branch Retinal Vein Occlusion] COMINO [A Phase III, Multicenter, Randomized, Double-Masked, Active Comparator-Controlled Study to Evaluate the Efficacy and Safety of Faricimab in Patients With Macular Edema Secondary to Central Retinal or Hemiretinal Vein Occlusion] |
Phase III, multicenter, randomized, double-masked, active comparator-controlled, parallel-group study in treatment-naïve patients with macular edema due to RVO |
BALATON: Aflibercept 2.0 mg (n=277) Faricimab 6.0 mg (n=276) COMINO: Aflibercept 2.0 mg (n=363) Faricimab 6.0 mg (n=366) |
Change from baseline in BCVA at week 24 |
At week 24 results were comparable between the two arms in each study.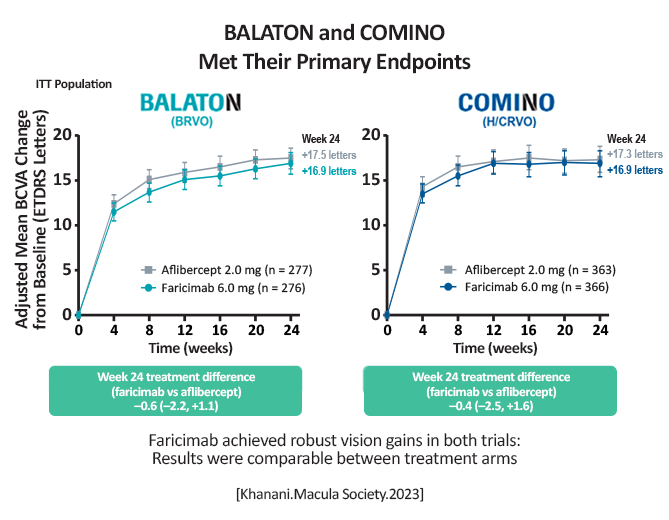
[Khanani.Macula Society.2023] BALATON: Faricimab: +16.9 letters; -311.4µm in central subfield thickness (CST) Aflibercept: +17.5 letters; -304.4µm in CST COMINO: Faricimab: +16.9 letters; -461.6µm in CST Aflibercept: +17.3 letters; -448.8µm in CST More patients achieved an absence of macular leakage with faricimab vs. aflibercept: BALATON, 33.6% vs. 21%, respectively (P = 0.0023) COMINO, 44.4% vs. 30%, respectively (P = 0.0002) [Khanani.Macula Society.2023] |
Faricimab met its primary endpoint and demonstrated a trend for advantages in absence of macular leakage. Faricimab was well tolerated, with a similar safety profile to aflibercept. [Khanani.Macula Society.2023] |
Faricimab is the first molecule to target 2 signaling pathways via the inhibition of angiopoietin-2 (Ang-2) and VEGF-A. |
|
BEACON [A Prospective, Randomized, Double-masked, Active Comparator-controlled, Multi-center, Two-arm, Phase 3 Study to Evaluate the Efficacy and Safety of Intravitreal KSI-301 Compared With Intravitreal Aflibercept in Participants With Visual Impairment Due to Treatment-naïve Macular Edema Secondary to Retinal Vein Occlusion (RVO)] |
Phase 3, prospective, randomized, double-masked, two-arm, multi-center non-inferiority study evaluating the efficacy and safety of repeated intravitreal dosing of tarcocimab 5 mg compared to aflibercept 2 mg in participants with visual impairment due to treatment-naïve macular edema secondary to RVO (either branch or central type). |
Tarcocimab dosed every 8 weeks Aflibercept dosed every 4 weeks |
Primary outcome was non-inferiority in change in BCVA from baseline to week 24 Tarcocimab: BRVO, n=220 CRVO, n=64 Aflibercept: BRVO, n=218 CRVO, n=66 |
At week 24, mean BCVA improved by 10.3 ETDRS letters in the tarcocimab group and by 11.2 ETDRS letters in the aflibercept group in BRVO.  [Shah.Retina Society.2023] Similar outcomes were reported for “All RVO”  [Shah.Retina Society.2023] Outcomes remained similar between the two groups from week 24 to week 48 Common ocular AEs were low and comparable between groups |
Tarcocimab treated patients had a ~30% higher chance of not requiring any additional doses versus aflibercept, even after receiving 2 fewer initiating doses (4 vs 6, respectively). Treatment burden distribution through 48 weeks had minimal overlap favoring tarcocimab, with 80% of tarcocimab patients receiving 5 or fewer doses vs 93% of aflibercept patients receiving 6 or more doses over 1 year After only 4 initiating doses in the first 6 months, approximately half of tarcocimab-treated patients required no additional injections through 12 months of treatment. [Shah.Retina Society.2023] |
Tarcocimab is an anti-VEGF antibody biopolymer conjugate (ABC) that blocks all VEGF-A isoforms. It may provide a viable option to currently marketed anti-VEGF treatments as BEACON showed it has similar outcomes to aflibercept, but with a much longer duration of treatment. |
| BRAVO, (RanibizumaB for the Treatment of Macular Edema following BRAnch Retinal Vein Occlusion) | Prospective, randomized, sham injection-controlled, double-masked, multicenter trial | Ranibizumab 0.3mg (n-134) Ranibizumab 0.5 mg (n=131) Sham (n=132) | Mean change from baseline BCVA letter score at month 6 | The mean change from baseline BCVA letter score was 16.6 in the 0.3 mg ranibizumab group, 18.3 in the 0.5 mg ranibizumab group, and 7.3 in the sham group (P<0.0001 for each ranibizumab group vs sham). | Monthly intravitreal ranibizumab injections provided important benefits for patients with macular edema following RVO, but several important questions remained, notably what percentage of patients would remain edema-free after ranibizumab treatment discontinuation as well as the impact of delays or gaps in treatment. [Campochiaro.2010] As-needed injections of ranibizumab maintained the improvement in visual acuity and central foveal thickness seen at month 6. | This was the first study to demonstrate anti-VEGF as an alternative to laser treatment for BRVOs with macular edema. The study design did not randomize patients to laser on day 0 so there is no direct head-to-head comparison. Nevertheless, treatment with laser resulted in suboptimal visual outcomes even when the patients were transitioned to anti-VEGF implying the 6 month delay in treatment is a significant concern. [Brown.2011] |
| BRVO (Comparing the Efficacy of Bevacizumab and Ranibizumab in Patients with Retinal Vein Occlusion) | Comparative, randomized, double-masked, multicenter, noninferiority clinical trial. | Bevacizumab 1.25 mg (n=144) Ranibizumab 0.5 mg (n-142) 133 diagnosed with BRVO 97 diagnosed with CRVO 47 diagnosed with hemi-CRVO | Change in BCVA of the study eye from baseline to 6 months, with a noninferiority margin of 4 letters. |
At 6 months, overall: BRVO Bevacizumab : 14.2 letter gain Bevacizumab : 16.1 letter gain |
Monthly administration of bevacizumab 1.25 mg is noninferior to monthly administration of ranibizumab 0.5 mg through month 6 in the treatment of patients with macular edema resulting from RVO. [Vader.2020] |
Patients in this study were treated monthly for 6 months, which is more than most real-world clinics will do. However, this adds to the literature and re-confirms noninferiority between bevacizumab and ranibizumab. |
| BVOS (Branch Vein Occlusion Study) | Multicenter, randomized trial | Group 1 included eyes at risk for the development of neovascularization, [BVOS.1984] group 2 included eyes at risk for the development of vitreous hemorrhage, [BVOS.1984] and group 3 included eyes at risk for vision loss from macular edema. [BVOS.1984] Group 3: Grid laser (n=71) No laser (n=68) |
For group 3, efficacy of macular grid laser for the treatment of macular edema secondary to BRVO
(Of note, there were no specific primary endpoints) |
At 3 years, the efficacy of laser treatment was clear; treated eyes in group 3 gained an average of 1.33 lines compared with 0.23 lines in a control group. [BVOS.1984] | Argon laser photocoagulation improved VA in patients with 20/40 or worse. Study authors recommended the treatment for those with VA reduced as a result of macular edema, not for those whose vision loss was a result of intraretinal hemorrhage in the fovea or fovea capillary nonperfusion. [BVOS.AJO.1984/p281/col1/para3] | Based on these data, macular grid laser photocoagulation became the first-line treatment for patients with macular edema following RVO. Laser treatment is considered a second-line treatment today only if patients are contraindicated for an anti-VEGF or if they are unlikely to keep up with monthly treatments. [Flaxel.2020] |
| Gutenberg Health Study [Incidence of retinal vein occlusion and its association with mortality - results from the Gutenberg Health Study] | Observational, population-based, prospective, single center cohort study in Europe | n/a, observational study | 5-year and 10-year cumulative incidence of BRVO and CRVO |
Cumulative incidence: BRVO: 5 y = 0.35% (95% CI: 0.25-0.5%; n=33) 10 y = 0.64% (95% CI: 0.46-0.87%; n=55) CRVO: 5 y = 0.043% (95% CI: 0.014-0.12%; n=4) 10 y = .14% (95% CI: 0.067-0.27%; n=12) 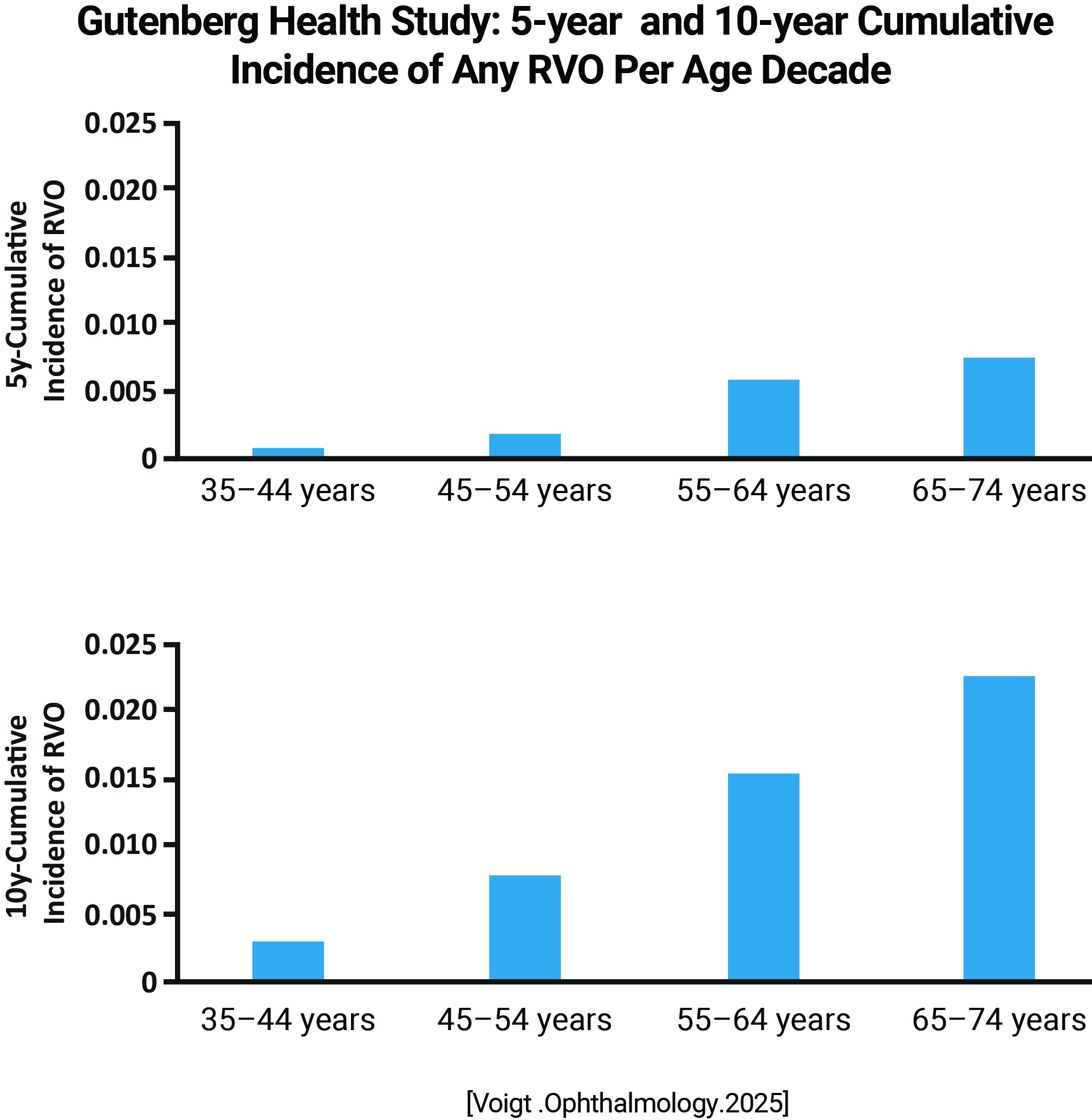
|
Incidence of retinal vein occlusion was associated with higher age There was an increased risk for mortality in subjects with RVO after adjustment for cardiovascular risk factors |
This study provides valuable data on RVO, their risk factors and associations These outcomes support the lower prevalence of RVO in Europe compared to the United States or Western pacific region reported elsewhere The increased risk for mortality was still significant after adjusting for major cardiovascular risk factors, highlighting the importance of managing cardiovascular risk factors in these patients. |
| QUASAR [Aflibercept 8 mg in Retinal Vein Occlusion: Primary Endpoint Results from the QUASAR study] | Multicenter, randomized, double-masked study in patients with treatment-naive macular edema secondary to RVO |
Aflibercept 2 mg Q4W Aflibercept 8 mg Q8W after 3 monthly loading doses Aflibercept 8 mg Q8W after 5 monthly loading doses |
Primary: Change from baseline in BCVA measured by the ETDRS letter score at Week 36 Secondary: Number of active injections from baseline Change from baseline in CRT |
At week 36, mean change from baseline in BCVA (ETDRS): 2Q4 = 17.5 8Q8/3 = 17.4 8Q8/5 = 18.3 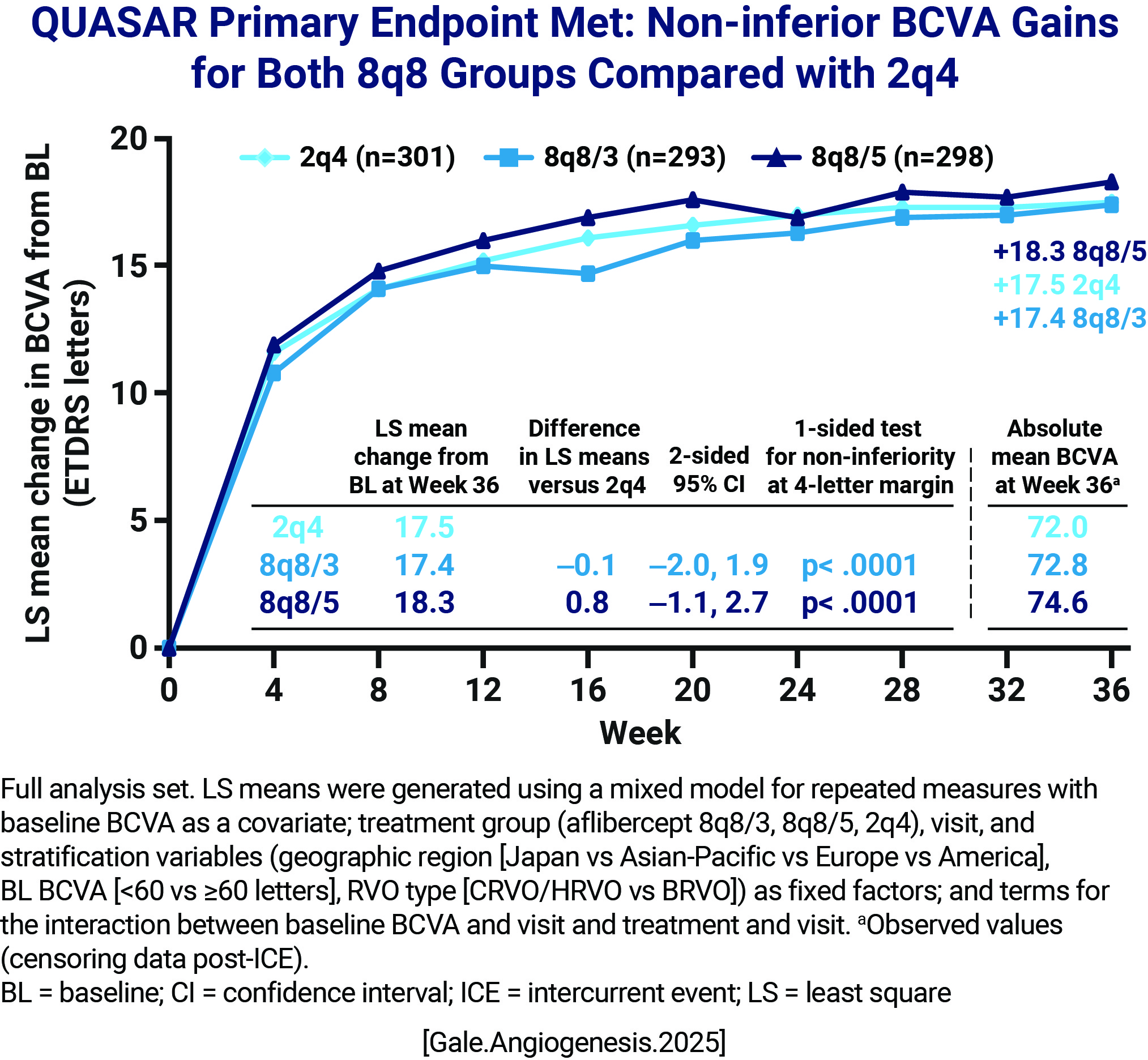 At week 36, last assigned dosing interval (Mean number of active injections): 2Q4 = 8.5 8Q8/3 = 6.0 8Q8/5 = 6.7 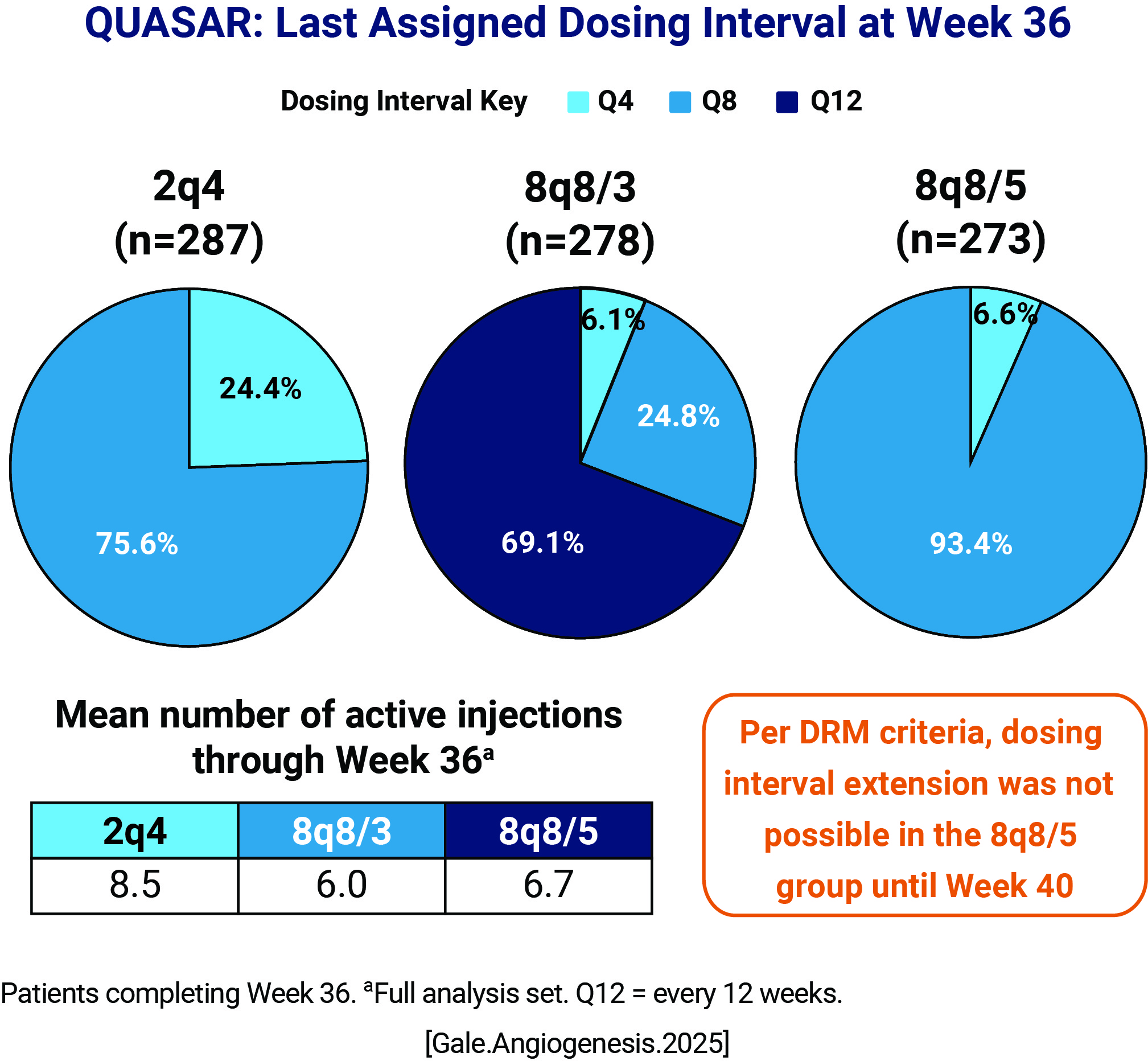 Per DRM criteria, dosing interval extension was not possible in the 8q8/5 group until Week 40 |
Aflibercept 8 mg groups achieved robust visual outcomes and non-inferior BCVA gains with fewer injections compared with the aflibercept 2 mg group at Week 36 Most patients in the aflibercept 8 mg groups maintained their assigned Q8 intervals through Week 36 Early, robust reductions in CRT were maintained through Week 36 Aflibercept 8 mg was well-tolerated, consistent with the established safety of aflibercept 2 mg, in patients with macular edema secondary to RVO, with no new safety signals |
Higher-dose aflibercept (8 mg compared to the traditional 2 mg) is equally efficacious at improving BCVA in RVO patients, but with a longer dosing interval For clinicians, this may mean a reduction in clinic treatment burden, and for patients it may mean fewer injections per year |
| VIBRANT [Study to Assess the Clinical Efficacy and Safety of Intravitreal Aflibercept Injection (IAI;EYLEA;BAY86-5321) in Patients With Branch Retinal Vein Occlusion (BRVO) | Phase 3, multicenter, randomized, double-masked, active-controlled trial | Macular grid laser photocoagulation (n=92) Aflibercept 2mg Q4 weeks (n=91) |
Treatment to week 20 Primary endpoint: proportion of patients 15 letters in BCVA from baseline [Campochiaro.2015] |
Week 24: Macular grid laser photocoagulation: 26.7% Aflibercept 2mg Q4 weeks: 52.7% [Campochiaro.2015] Week 52: Macular grid laser photocoagulation: 41.1% Aflibercept 2mg Q4 weeks: 57.1% [Clark.2016] | Treatment with aflibercept 2 mg every 4 weeks was superior to laser treatment through week 24 in visual acuity gains and anatomic outcomes. [Clark.2016] Continued treatment with an every 8-week regimen maintained visual and anatomic gains through week 52 after 6 monthly injections. [Clark.2016] | The study randomized patients in a head-to-head fashion on day 0 to either laser or aflibercept treatment.The results further validated that anti-VEGF treatment is superior to laser treatment. [Clark.2016] |